 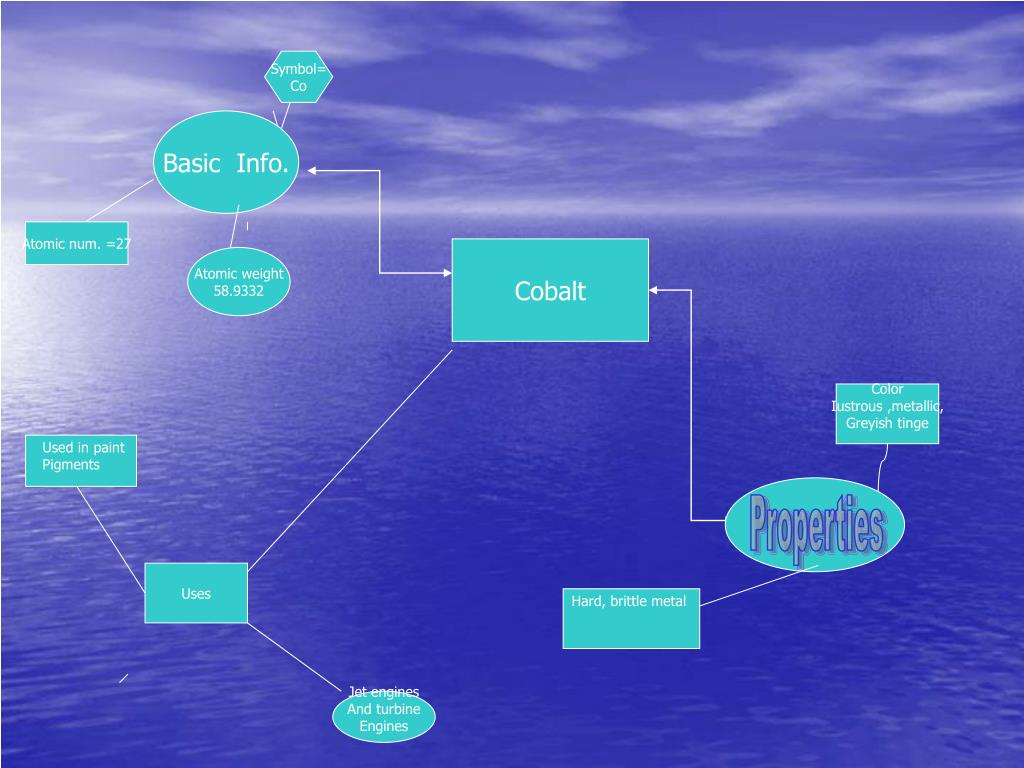
The compound can be prepared by reacting cobalt(II) nitrate in water with a solution of triethylamine N(Cģ as both the base and a complexing agent. Ĭobalt(II) hydroxide is most used as a drying agent for paints, varnishes, and inks, in the preparation of other cobalt compounds, as a catalyst and in the manufacture of battery electrodes.Ĭobalt(II) hydroxide precipitates as a solid when an alkali metal hydroxide is added to an aqueous solution of Co 2+ salt. This compound is blue and rather unstable. The name is also applied to a related compound, often called "alpha" or "blue" form (α- Co(OH)Ģ), which incorporates other anions in its molecular structure. The pure compound, often called the "beta form" (β- Co(OH)Ģ) is a pink solid insoluble in water. Cobalt is not known to bio-magnify in animal food chain hence if the herbivorous animal gets eaten by carnivorous animal then the cobalt won’t increase in the body of the carnivorous animal.Cobalt(II) hydroxide or cobaltous hydroxide is the inorganic compound with the formula Co(OH)Ģ, consisting of divalent cobalt cations Co 2+ Animals: Herbivorous animals may eat the cobalt-rich plants which lead to excess of cobalt inside their body.It leads to competition of absorption of nutrients which may lead to excess of cobalt in plants and deficiency of some other mineral. Since this cobalt is not bound to soil, it easily gets absorbed by the plants. Plants: Cobalt released from industries via combustion action into the atmosphere gets settled on the soil.Eating such fruits regularly can cause over-uptake of cobalt and excess of cobalt in human body which leads to nausea, vomiting and problems in heart, thyroid, and eyes. Excess cobalt usually gets stored in fruits and seeds. Over-uptake: If cobalt is not bound to the soil or sedimentary particles then it gets easily absorbed by crops and plants.Inhalation: If cobalt is inhaled in large amounts which can happen at cobalt centered work places then it causes asthma and pneumonia.Cobalt is also used in nickel-cadmium and nickel metal hydride batteries to improve oxidation of nickel. They are mixed with chromium, tungsten, molybdenum, nickel, aluminium, iron, etc and used in turbines, aircrafts, orthopedic equipments, jewellery etc.īatteries: Lithium cobalt oxide is used in lithium-ion battery cathodes to release lithium ions during operation. Superalloys: The cobalt metal is mixed with many metals to create high performance alloys called superalloys which are resistant to high temperatures, corrosion, and wear.They are used in Fisher-Tropsch process, Hydroformylation of alkene, to make terepthalic acid, to make paints, varnishes, as drying agents, to accelerate growth of polyester resins, in hydrodesulfurization of petroleum, etc. Catalysts: Cobalt compound based catalysts are widely used in chemical and related industries.Relevance in Chemical and Related Industries Aluminothermic reaction: In order to obtain metallic cobalt, the products obtained from the above processes is oxidised to cobalt oxide and then the metallic cobalt is recovered using aluminothermic reaction.The residue is further leached with sulphuric acid. Water is used as a leaching solvent to extract sulphates and arsenates. Leaching: The cobalt sulphate solutions may be leached fur further refining.The cobalt sulphate has many applications and can be sold directly in market. The enriched ore is then roasted to oxidise copper and iron to its subsequent oxides and obtain cobalt sulphate solution. The surfactants get bound to components of the ore in such a way that it leads to enrichment of the cobalt metal in the ore. Froth flotation: The ore is subjected to surfactants.Cobalt reacts with all the halogens at high temperatures to yield binary halides.Cobalt easily gets attacked by halogens and sulfur.Metallic cobalt is weakly reducing in nature.Cobalt reacts slowly with mineral acids.Cobalt reacts very slowly with the moist air at ambient conditions it does not react with dry air.If cobalt is exposed to air then a passivation oxide layer forms on the surface of the metal.There are 22 radioisotopes of cobalt which has been characterized till now. Cobalt-59 is the only stable isotope of cobalt which is naturally found on earth.Metallic cobalt occurs in two structures face-centred cubic (fcc) and hexagonal close packing (hcp).Cobalt is a silvery-blue lustrous metal which is magnetic in nature.Several tonnes of cobalt are also found in deepest parts of the ocean in manganese rich nodules these nodules contain many transition metals. The most common minerals which contain cobalt are cobaltite, skutterudite, and erythrite. It makes up to 0.0029% of the Earth’s crust. Cobalt is found in nature in combined form except for very small deposits of free cobalt found in alloys of natural meteoric iron. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
